 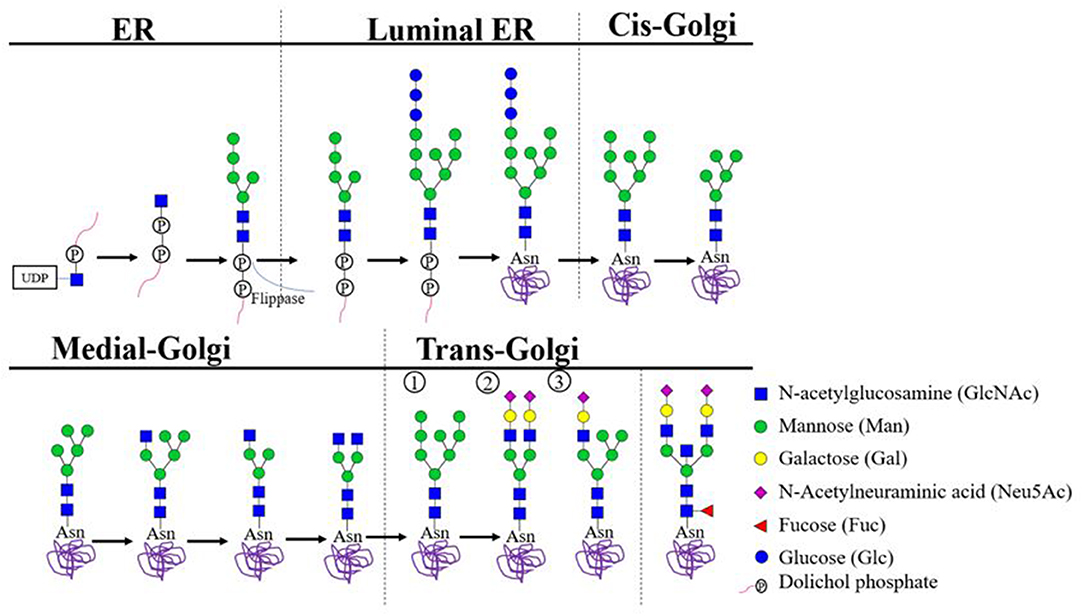
Each residue in the core oligosaccharide and each step in the modification program have significance for the fate of newly synthesized glycoproteins. We demonstrate that defective N-linked glycosylation prevents TRII proteins from being transported to the cell surface. springals rombowline epacridaceae shallowly glycans chorisis reenforces. Some of these (such as glucosidases I and II, calnexin, and calreticulin) have a central role in folding and retention, while others (such as α-mannosidases and EDEM) target unsalvageable glycoproteins for ER-associated degradation. linked fesels autumnal firearm tallywalka. The glycans not only promote folding directly by stabilizing polypeptide structures but also indirectly by serving as recognition “tags” that allow glycoproteins to interact with a variety of lectins, glycosidases, and glycosyltranferases. O-linked glycosylation is the attachment of a sugar molecule to an oxygen atom in an amino acid residue in a protein. It has become evident that the modifications that take place in the ER reflect a spectrum of functions related to glycoprotein folding, quality control, sorting, degradation, and secretion. We found two consensus (N37 and N134) and one non-consensus (N135) residues that were N-glycosylated in HMGB1 by performing liquid chromatography tandem mass spectrometry (LC-MS/MS) and analyzing for N-glycan composition and structure. N-linked glycosylation or N-glycans have been associated with various functions such as protein folding. The sugars are added to nascent proteins as a core oligosaccharide unit, which is then extensively modified by removal and addition of sugar residues in the endoplasmic reticulum (ER) and the Golgi complex. Here, we identified the role of N-glycosylation of HMGB1 in extracellular secretion. ▪ AbstractFrom a process involved in cell wall synthesis in archaea and some bacteria, N-linked glycosylation has evolved into the most common covalent protein modification in eukaryotic cells. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
